
After enough mRNA has been produced in an IVT reaction, the pDNA is broken down to ensure that only mRNA will be incorporated into the vaccine.īecause mRNA is highly susceptible to degradation, a lipid nanoparticle (LNP) or other suitable delivery system must be developed to package the mRNA and deliver it to cells. Additional steps are performed using distinctive enzymes to cap and thus stabilize that material. Plasmids easily replicate and magnify target gene sequences, enabling rapid production.įigure 1: Manufacturing process for mRNA-based vaccines (DS = drug substance, DP = drug product).ĭuring a cell-free in vitro transcription (IVT) process, a pDNA template is incubated with enzymes and nucleotides to produce mRNA. The resulting plasmid DNA (pDNA) undergoes purification and linearization. Inducing antibody binding with the S protein would prevent attachment of a virus particle to human cells exhibiting the angiotensin-converting enzyme 2 (ACE-2) receptor, thereby preventing viral entry.Īfter target identification, scientists insert the DNA sequence coding for the gene of interest (GoI) (for SARS-CoV-2 vaccines, that is the gene encoding for the S protein) into a plasmid, which is amplified in host bacteria (typically Escherichia coli) using a fermentor (Figure 1). Hence, in the case of COVID-19 vaccines, many drug companies selected the SARS-CoV-2 S protein. However, the target protein must be sufficiently different from proteins in human cells so that resulting immune responses detect and address only the pathogen. The outcome has been rapid availability of COVID-19 mRNA vaccines in major markets, based on work from manufacturing resources that are spread out around the world.ĭuring mRNA vaccine development, scientists first must identify a target protein from the pathogen of interest - e.g., in the case of SARS-CoV-2, the spike (S) protein. However, unprecedented global collaboration, funding, and government support relating to the COVID-19 pandemic accelerated efforts to address the difficulties surrounding mRNA technology. The modality did not gather momentum during early studies because of immunogenicity, stability, and delivery issues. Scientists have been studying mRNA vaccines for almost three decades to protect against influenza, rabies, Zika virus, and cytomegalovirus (CMV) ( 3). Pfizer–BioNTech’s and Moderna’s respective vaccines were developed in such short timeframes because mRNA technology was not designed overnight to mitigate COVID-19. The rapidity with which those products were developed stands in stark contrast to the 10–15 years that traditionally have been required for a candidate vaccine to move from discovery to approval and administration in patients. 
EUA for Moderna’s mRNA vaccine followed soon after. Within a couple months of the virus’s gene sequence being published ( 1), Pfizer and BioNTech were ready with their vaccine candidate ( 2), and in less than a year, the vaccine was approved for administration in adults. Kate Stegeman, an advocacy coordinator for International aid group Medicins Sans Frontieres, called the announcement "a welcome milestone".Vaccines based on messenger ribonucleic acid (mRNA) created headlines in December 2020 for being the first highly efficacious SARS-CoV-2 prophylactics to receive emergency use authorization (EUA) from the US Food and Drug Administration (FDA). To date, over 20 countries have requested access to the hub’s technology transfer, the WHO said. The mRNA hub in South Africa has a global approach, serving not only Africa but the world. 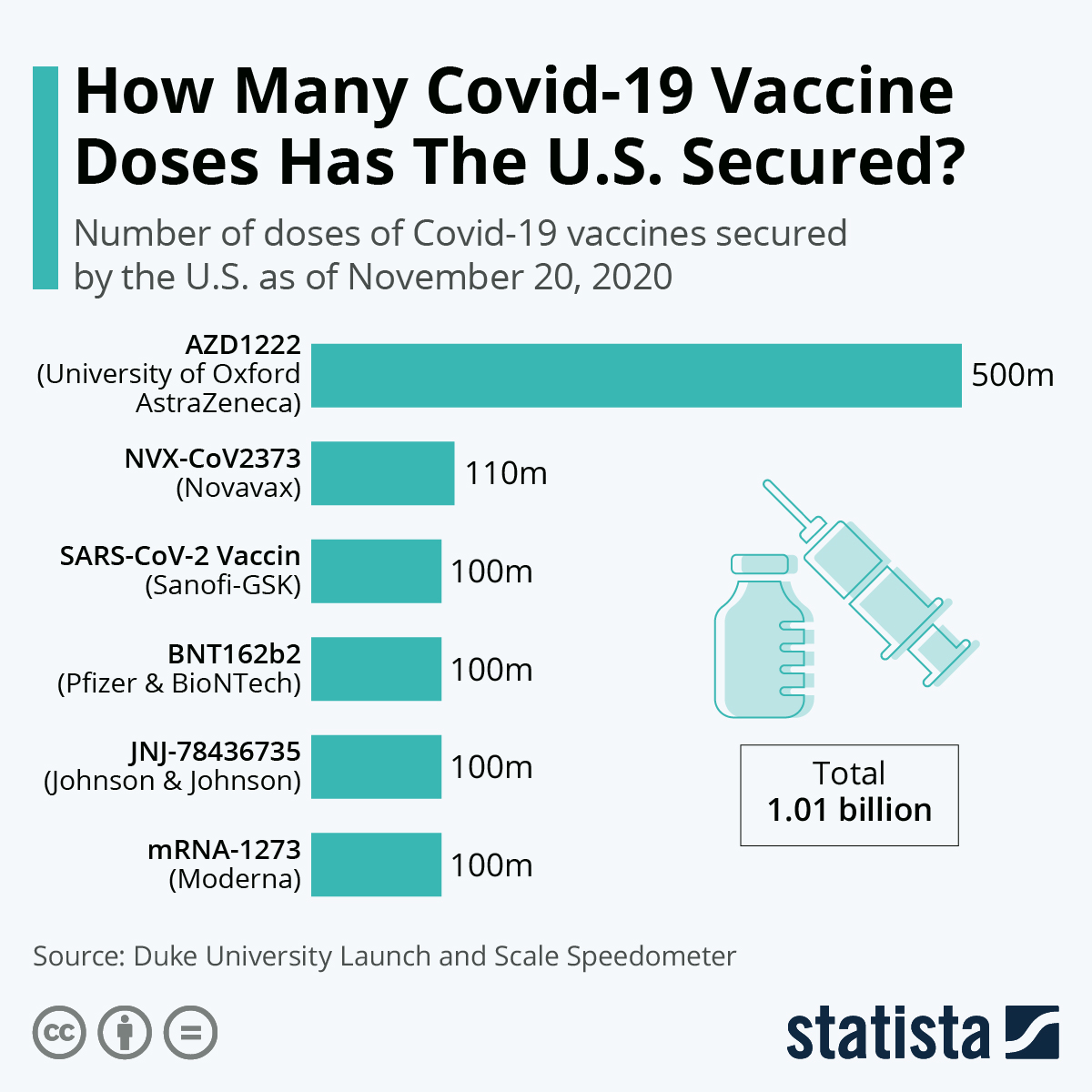 
Other manufacturing "spokes" in the WHO's hub-and-spoke concept include Argentina and Brazil. The first recipient of the mRNA technology transfer is consortium partner and partly state-owned South African vaccine manufacturer Biovac, which will mass produce the vaccine once it has passed the necessary safety and regulatory hurdles. Primarily set up in response to the COVID-19 emergency, the transfer hub could expand manufacturing capacity to tackle diseases such as tuberculosis and malaria in Africa. "This is mRNA technology designed in Africa, led by Africa and owned by Africa, with the support of Team Europe," European Commission President Ursula von der Leyen said. Training of the recipient countries will begin in March 2022. The hub has already established mRNA vaccine production at laboratory scale and is working towards commercial production. Makolo did not respond directly on whether Rwanda had applied to be a part of the WHO’s technology transfer project. Rwandan government spokeswoman Yolande Makolo told Reuters the government is working with BioNTech, the WHO and the AU on mRNA vaccine manufacturing, citing an agreement BionTech signed with Kigali and Senegal's Institut Pasteur of Dakar in October. Senegalese President Macky Sall said: "Our aim of course is to have 60% of vaccines given in Africa.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
